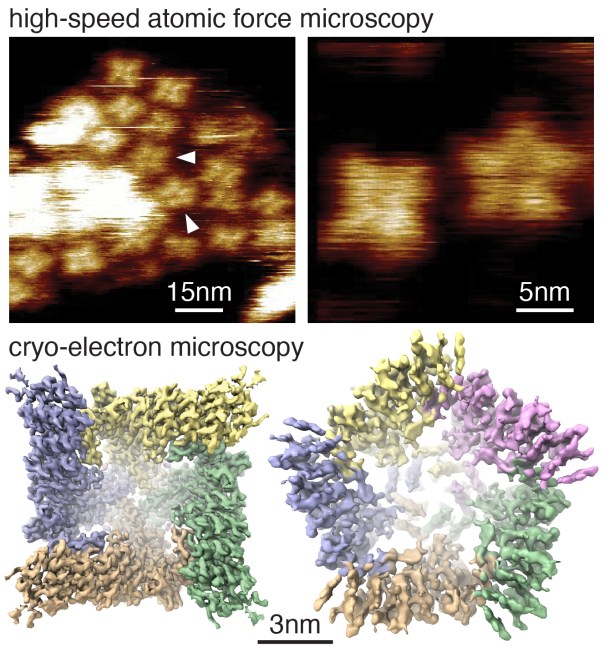
The Scheuring-Lab / Bio-AFM-Lab is specialized in Atomic Force Microscopy (AFM) based technologies for the study of various membrane phenomena, such as membrane protein structure, assembly, diffusion and conformational dynamics. Over the past few years, my laboratory has been instrumental in the development and application of High-Speed Atomic Force Microscopy (HS-AFM), unique for the analysis of dynamics of unlabeled single molecules, allowing to bridge structure and function. Using HS-AFM, we reach ~1nm lateral, ~0.1nm vertical and ~100ms temporal resolution. While we continuously push the technological limits, our state-of-the-art HS-AFM can readily resolve dynamics of single domains in transmembrane channels, transporters and membrane associated proteins.
To make HS-AFM more powerful for biological applications, we developed environmental control, i.e. slow and fast buffer exchange, temperature and force control. We further developed novel AFM-based modalities, by integrating optical microscopy into a HS-AFM, and by developing high-speed force spectroscopy (HS-FS) and high frequency microrheology (HF-µR). We also developed HS-AFM line scanning (HS-AFM-LS) and HS-AFM height spectroscopy (HS-AFM-HS) which allows to probe molecular dynamics experimentally at millisecond and microsecond temporal resolution. Most recently, we introduced localization atomic force microscopy (LAFM) a super-resolution method that allows the extraction of quasi-atomic structural details from single molecule AFM data.
Complementary to HS-AFM imaging that can uncover the structural dynamics of membrane proteins at work, we apply cryogenic electron microscopy (cryo-EM0 to solve atomic structures of membrane proteins. The combined understanding of high-resolution structures, conformational variability, and single-molecule kinetics provide unprecedented insights into membrane protein structure and function. Our microscopy efforts are often further complemented by computational investigations, and other biophysical methods.